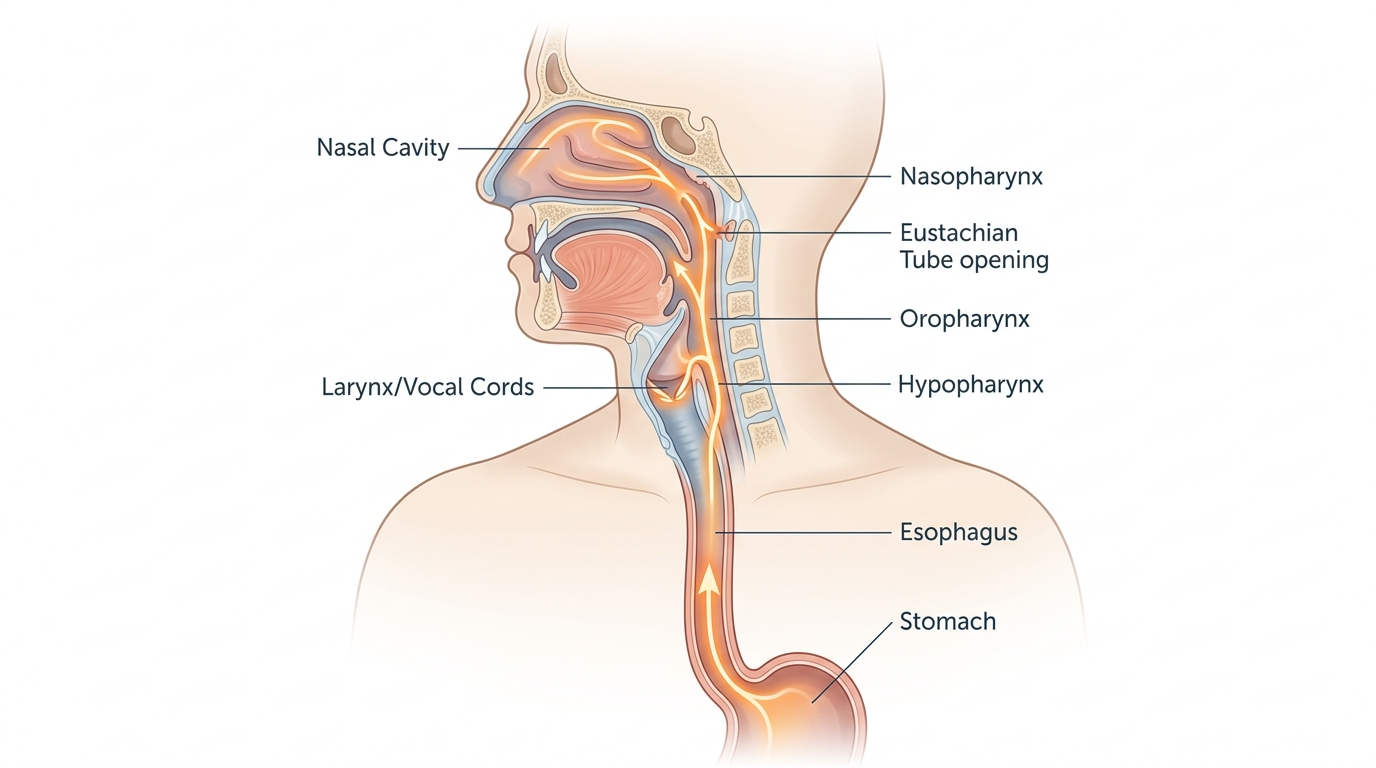
Acid reflux and anxiety can feel similar because both can cause chest discomfort, throat tightness, nausea, and a lump-in-the-throat sensation. The main difference is pattern. Reflux symptoms usually track with meals, bending, or lying down. Anxiety symptoms usually track with stress, racing thoughts, palpitations, or sudden episodes that are not clearly meal-linked.
How we evaluated acid reflux versus anxiety symptoms
We prioritized symptom-pattern guidance from the Mayo Clinic GERD overview, the Cleveland Clinic reflux overview, and National Institute of Mental Health resources on anxiety disorders and panic symptoms. We compared timing, triggers, and associated symptoms rather than treating one symptom in isolation. We excluded disease diagnosis advice because chest pain, weight loss, vomiting, and swallowing problems need clinician review.
Why do acid reflux and anxiety get confused so often?
Acid reflux and anxiety overlap because both systems recruit the chest, throat, and upper abdomen. Reflux can create heartburn, sour taste, throat clearing, hoarseness, nausea, and a pressure sensation behind the breastbone, as described by the Mayo Clinic. Anxiety can create chest tightness, shortness of breath, nausea, stomach discomfort, dizziness, and a sense that something is very wrong, which the NIMH panic disorder guide lists as common physical symptoms. The vagus nerve and stress hormones make the overlap worse because gut sensation becomes louder when the nervous system is activated. That means a person may feel burning and panic at the same time, or may feel anxiety because the reflux sensation itself is alarming. Symptom overlap is real, but the body still leaves clues if you stop looking at a single moment and start looking at the full pattern.
What clues point more strongly toward acid reflux than anxiety?
Acid reflux usually follows mechanics and meal timing. Symptoms often appear after large meals, fatty foods, alcohol, late-night eating, bending forward, or lying flat. Reflux also tends to bring a sour taste, throat irritation, chronic throat clearing, regurgitation, or burning that rises upward, which are features emphasized by the Cleveland Clinic. Night symptoms matter too. If discomfort is worse after dinner or wakes you from sleep with a bitter taste or cough, reflux moves higher on the list. Reflux can happen without classic heartburn, especially when throat symptoms dominate. Food relationships are the biggest tell. Anxiety episodes can happen after meals, but reflux is much more likely to repeat after the same physical triggers. A simple symptom log that tracks meal size, body position, and timing often reveals a clearer reflux pattern within a week than memory alone can provide.
What clues point more strongly toward anxiety than acid reflux?
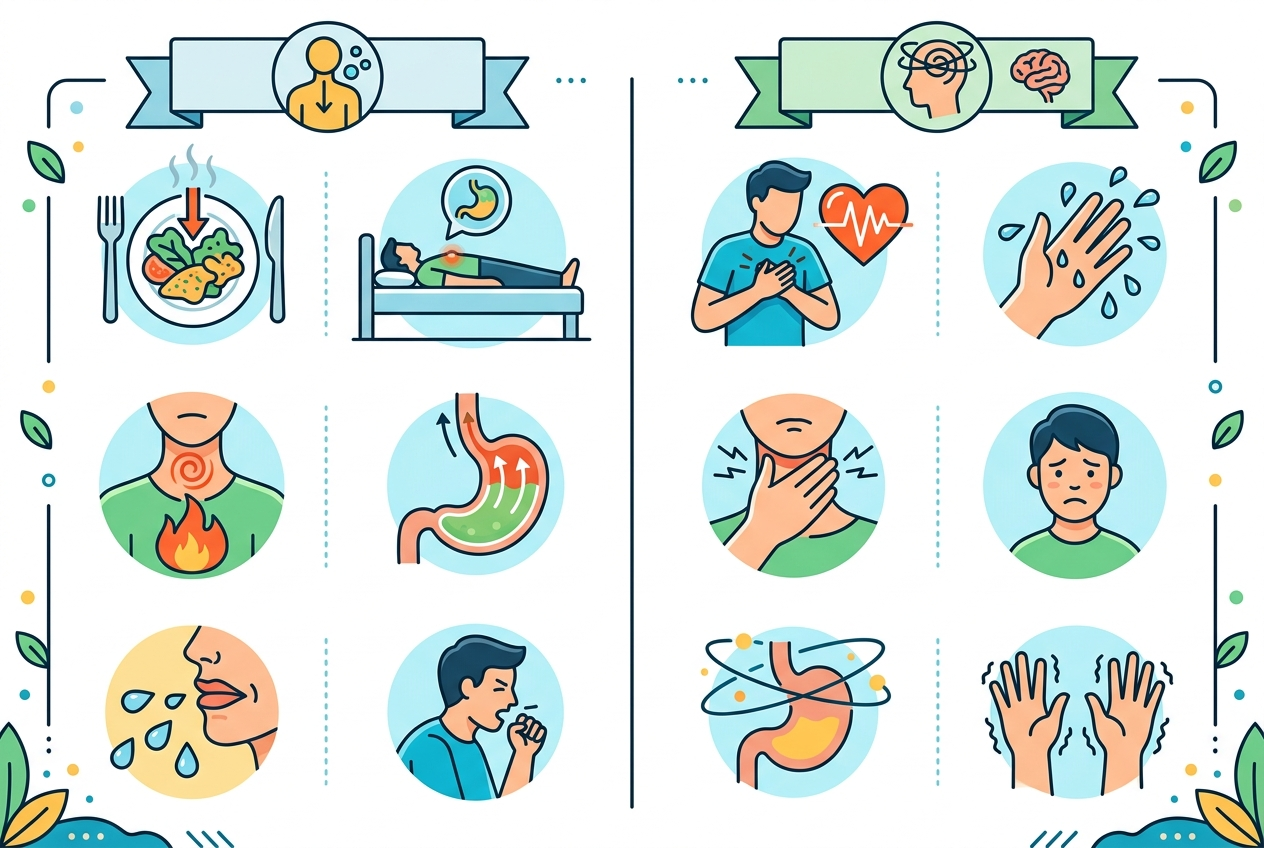
Anxiety usually follows nervous-system activation more than food exposure. Symptoms often rise during conflict, overthinking, deadline pressure, social stress, or sudden waves of fear. The NIMH anxiety disorders page notes that anxiety can produce restlessness, muscle tension, rapid heartbeat, sleep disruption, stomach upset, and trouble concentrating. When chest discomfort arrives with shaking, sweating, tingling, racing thoughts, dread, or a sense of losing control, anxiety becomes more likely than reflux alone. Anxiety symptoms can also move around the body in ways reflux does not. One episode might feel like chest pressure, another like nausea, another like dizziness or throat tightness. That variability is common in anxiety states. Reflux is usually more mechanically consistent. It follows meal timing and body position. Anxiety follows context, anticipation, and internal alarm signals. The cleaner the stress link, the stronger the anxiety clue.
When should you stop guessing and get checked?
Self-patterning is useful, but some symptoms deserve medical evaluation instead of home detective work. Chest pain with exertion, shortness of breath that feels severe, black stool, vomiting blood, trouble swallowing, unexplained weight loss, persistent vomiting, or symptoms that keep worsening should not be written off as either reflux or anxiety. The Mayo Clinic and Cleveland Clinic both flag swallowing difficulty and unintentional weight loss as warning signs. The same logic applies if anxiety symptoms are frequent enough to limit sleep, work, or basic functioning. The NIMH notes that persistent anxiety disorders are treatable, but they need real assessment. Pattern recognition is helpful. It is not a substitute for urgent care when symptoms are intense, new, or paired with red flags.
For a detailed comparison of specific products and strains, see Gallbladder or GERD? How to Tell the Difference and What to Do About It.
FAQ
Can anxiety really cause nausea and throat tightness?
Yes. The NIMH describes stomach pain, nausea, chest discomfort, and breathing-related sensations as common physical symptoms during panic and anxiety episodes. Those symptoms can feel very similar to upper digestive discomfort.
Can reflux happen without obvious heartburn?
Yes. Some people have throat clearing, cough, hoarseness, nausea, or regurgitation without a classic burning feeling. That is one reason reflux and anxiety get mixed up so often.
Is meal timing the most useful reflux clue?
Usually yes. Symptoms that repeat after meals, late at night, or when lying down point more strongly toward reflux than anxiety. Stress can still worsen the experience, but timing gives reflux away.
Does stress make reflux worse even if reflux is the main issue?
Yes. Stress does not create stomach acid out of nowhere, but it can amplify gut sensitivity and make ordinary reflux feel sharper. Mixed patterns are common.
Should you track symptoms for a few days before deciding?
Yes. A short log of meals, body position, stress level, and associated symptoms is often more useful than guessing from memory. Patterns show up fast when they are written down.
When is chest discomfort an emergency?
If chest pain is severe, comes with shortness of breath, fainting, sweating, arm or jaw pain, or feels different from your usual symptoms, it deserves urgent medical care. That is not the time for home sorting.